Prescription Lidocaine Patches, HCG Prescription, and Prescription Orthotics Guide
Prescription Lidocaine Patches, HCG Prescription, and Prescription Orthotics Guide
Prescription medications and devices that exist alongside over-the-counter alternatives often confuse patients about what requires a provider visit. Prescription lidocaine patches provide localized pain relief at concentrations not available without a prescription, and understanding when they are appropriate, how they differ from OTC lidocaine products, and what to expect from lidocaine patch prescription management helps patients get the most from this therapy. Similarly, hcg prescription uses span medical weight management and fertility treatment, making clarity about the clinical context essential. And prescription orthotics, as distinct from OTC insoles, require specific clinical justification.
Each of these prescription products occupies a specific clinical niche where provider evaluation, monitoring, and individualized prescription are justified. Understanding why these products are prescription-only, rather than simply following instructions on a label, helps patients engage more meaningfully with their providers about whether these options are appropriate for their situations.
Prescription Lidocaine Patches: What They Are and How They Work
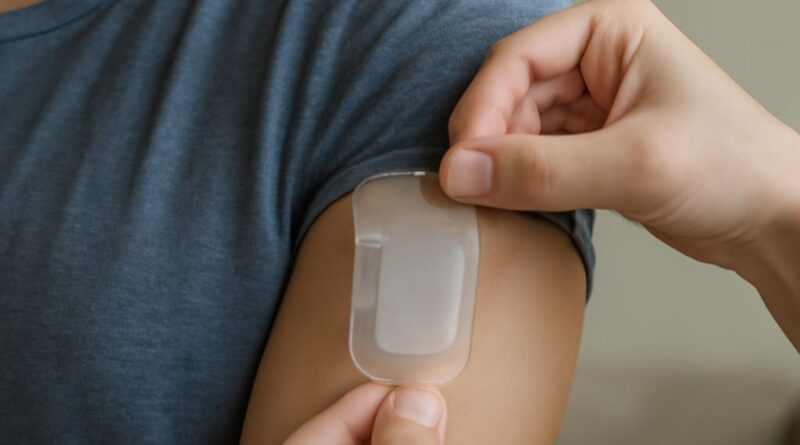
Prescription lidocaine patches are available in a 5 percent concentration, significantly higher than the OTC 4 percent maximum. The most commonly prescribed formulation is the Lidoderm patch, available generically as lidocaine patch 5 percent. These patches deliver localized anesthesia to skin and superficial tissues in the application area, providing pain relief for conditions including postherpetic neuralgia (nerve pain following shingles), local nerve pain from injury or surgery, and in some cases localized musculoskeletal pain.
The lidocaine patch prescription dosing protocol is specific: up to three patches applied to the most painful area for up to 12 hours within a 24-hour period. The 12-hour on, 12-hour off schedule allows time for elimination of the small amount of lidocaine that is absorbed systemically and prevents accumulation. Patients applying more patches than prescribed or leaving them on longer than directed risk higher systemic lidocaine absorption, which can cause cardiac and neurological effects in susceptible individuals.
Prescription lidocaine patches should not be applied to broken or irritated skin. The area should be clean and dry before application, and used patches should be disposed of carefully because they retain a significant amount of active lidocaine even after use. Patients with allergies to amide-type local anesthetics should not use lidocaine patches. Drug interactions with antiarrhythmic medications are possible and should be disclosed to the prescribing provider and pharmacist.
HCG Prescription: Medical Uses and Regulatory Context
HCG (human chorionic gonadotropin) is a prescription hormone used in several clinical contexts. In fertility treatment, hcg injection triggers ovulation in women undergoing ovarian stimulation protocols and supports luteal phase function. In men, hcg is used to stimulate testosterone production in hypogonadism or to maintain testicular function and fertility during testosterone replacement therapy.
The hcg prescription for weight loss has a more complex regulatory history. The FDA has specifically stated that hcg is not approved for weight loss and that marketing hcg for weight loss is fraudulent. The extreme caloric restriction that typically accompanies hcg weight loss protocols, often 500 calories per day, produces weight loss through the caloric deficit rather than through any pharmacological effect of hcg. The FDA and FTC have issued warning letters to companies marketing hcg weight loss programs, and the evidence base for this use does not support clinical benefit attributable to hcg itself.
Patients who encounter hcg weight loss programs should be aware that these protocols are not FDA-approved for this indication and that the dramatic weight loss from severe caloric restriction carries its own health risks. Consulting with a physician or registered dietitian about evidence-based weight management strategies provides a safer and more sustainable alternative to extreme caloric restriction protocols regardless of what adjunctive medications are marketed alongside them.
Prescription Orthotics: When Custom Orthotics Are Clinically Justified
Prescription orthotics are custom-fabricated foot orthotic devices based on a clinical evaluation and a mold or digital scan of the patient foot. They differ from over-the-counter insoles in that they are made specifically for the patient foot shape and corrective requirements rather than for a general population. The clinical indications for prescription orthotics include biomechanically significant flat foot or high arch, plantar fasciitis that has not responded to conservative measures, posterior tibialis tendon dysfunction, and certain cases of knee, hip, or lower back pain with a documented biomechanical foot component.
Prescription orthotics are typically prescribed by podiatrists or orthopedic surgeons and fabricated by a certified orthotist or prosthetic and orthotic specialist. The fabrication process begins with a foot impression or digital scan taken in a neutral subtalar position. The impression is used to cast a custom device from rigid or semi-rigid polypropylene or similar material, sometimes with soft top covers for cushioning. The total cost of custom prescription orthotics ranges from $300 to $600 per pair at most practices.
Insurance coverage for prescription orthotics varies significantly. Medicare has specific coverage criteria for therapeutic shoes and inserts for diabetic patients with documented foot complications, but coverage for orthotics in non-diabetic patients is more limited. Many private insurance plans cover custom orthotics when prescribed by a physician for a qualifying diagnosis and when conservative treatment has been documented as insufficient. Obtaining prior authorization before ordering custom orthotics prevents unexpected out-of-pocket cost.
When Prescription Dosing Forms Matter: Patches, Injections, and Devices
The dosing form of a prescription product matters significantly for clinical outcomes. A prescription lidocaine patch delivers medication locally without meaningful systemic absorption in most patients, making it appropriate for patients who might not tolerate oral analgesics. An oral lidocaine equivalent does not exist for the same indication because systemic oral lidocaine administration for pain would require careful cardiac monitoring.
Similarly, hcg prescription uses require subcutaneous injection or intramuscular injection because the hormone is not effective when taken orally. Oral hcg products marketed over the counter are pharmacologically inactive because hcg is a protein that is digested and destroyed in the gastrointestinal tract before it can reach systemic circulation. Patients who receive oral hcg products are not receiving active medication regardless of the marketing claims on the packaging.
Prescription orthotics as a device category illustrate that not all prescription products are medications. Medical devices prescribed and custom-fabricated based on clinical evaluation occupy the same regulatory category in terms of requiring professional oversight, though the regulatory pathway differs from drug approval. Patients who receive recommendations for prescription-only dosing forms or devices should feel empowered to ask why this specific form was chosen and what alternatives exist, including both prescription and non-prescription options, before agreeing to the recommended treatment.